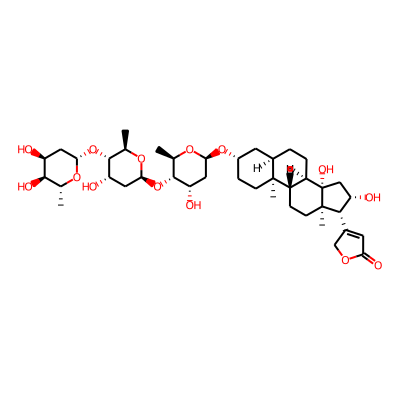
Summary
SMILES: O=C1OCC(=C1)[C@H]1[C@@H](O)C[C@]2([C@]1(C)CC[C@H]1[C@H]2CC[C@H]2[C@]1(C)CC[C@@H](C2)O[C@H]1C[C@H](O)[C@@H]([C@H](O1)C)O[C@H]1C[C@H](O)[C@@H]([C@H](O1)C)O[C@H]1C[C@H](O)[C@@H]([C@H](O1)C)O)OInChI: InChI=1S/C41H64O14/c1-19-36(47)27(42)14-33(50-19)54-38-21(3)52-34(16-29(38)44)55-37-20(2)51-32(15-28(37)43)53-24-8-10-39(4)23(13-24)6-7-26-25(39)9-11-40(5)35(22-12-31(46)49-18-22)30(45)17-41(26,40)48/h12,19-21,23-30,32-38,42-45,47-48H,6-11,13-18H2,1-5H3/t19-,20-,21-,23-,24+,25+,26-,27+,28+,29+,30+,32+,33+,34+,35+,36-,37-,38-,39+,40-,41+/m1/s1InChIKey: LKRDZKPBAOKJBT-CNPIRKNPSA-N
DeepSMILES: O=COCC=C5)[C@H][C@@H]O)C[C@][C@]5C)CC[C@H][C@H]6CC[C@H][C@]6C)CC[C@@H]C6)O[C@H]C[C@H]O)[C@@H][C@H]O6)C))O[C@H]C[C@H]O)[C@@H][C@H]O6)C))O[C@H]C[C@H]O)[C@@H][C@H]O6)C))O))))))))))))))))))))))))))))O
Scaffold Graph/Node/Bond level: O=C1C=C(C2CCC3C2CCC2C4CCC(OC5CCC(OC6CCC(OC7CCCCO7)CO6)CO5)CC4CCC23)CO1
Scaffold Graph/Node level: OC1CC(C2CCC3C2CCC2C4CCC(OC5CCC(OC6CCC(OC7CCCCO7)CO6)CO5)CC4CCC23)CO1
Scaffold Graph level: CC1CCC(C2CCC3C2CCC2C4CCC(CC5CCC(CC6CCC(CC7CCCCC7)CC6)CC5)CC4CCC23)C1
Functional groups: CC1=CC(=O)OC1; CO; C[C@H](OC)OC
Chemical classification
ClassyFire Kingdom: Organic compounds
ClassyFire Superclass: Lipids and lipid-like moleculesClassyFire Class: Steroids and steroid derivatives
ClassyFire Subclass: Steroid lactones
NP Classifier Biosynthetic pathway: Terpenoids
NP Classifier Superclass: Steroids
NP Classifier Class: Cardenolides
Synonymous chemical names:gitoxin
External chemical identifiers:CID:91540; ChEMBL:CHEMBL109317; ChEBI:28503; ZINC:ZINC000238809171; FDASRS:VOR2TD6SO4; SureChEMBL:SCHEMBL309847
Chemical structure download