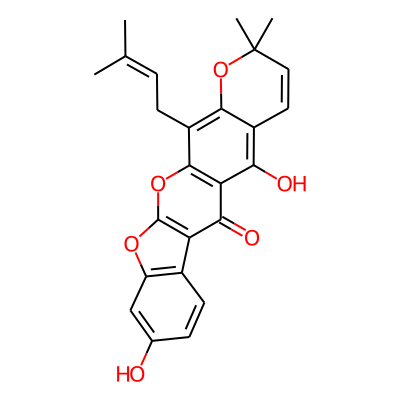
| Property name | Tool | Property value |
|---|
| Molecular weight (g/mol) | RDKit | 0 |
| Log P | RDKit | 0 |
| Topological polar surface area (Å2) | RDKit | |
| Number of hydrogen bond acceptors | RDKit | |
| Number of hydrogen bond donors | RDKit | |
| Number of carbon atoms | RDKit | |
| Number of heavy atoms | RDKit | |
| Number of heteroatoms | RDKit | |
| Number of nitrogen atoms | RDKit | |
| Number of sulfur atoms | RDKit | |
| Number of chiral carbon atoms | RDKit | |
| Stereochemical complexity | RDKit | 0 |
| Number of sp hybridized carbon atoms | RDKit | |
| Number of sp2 hybridized carbon atoms | RDKit | |
| Number of sp3 hybridized carbon atoms | RDKit | |
| Shape complexity | RDKit | |
| Number of rotatable bonds | RDKit | |
| Number of aliphatic carbocycles | RDKit | |
| Number of aliphatic heterocycles | RDKit | |
| Number of aliphatic rings | RDKit | |
| Number of aromatic carbocycles | RDKit | |
| Number of aromatic heterocycles | RDKit | |
| Number of aromatic rings | RDKit | |
| Total number of rings | RDKit | |
| Number of saturated carbocycles | RDKit | |
| Number of saturated heterocycles | RDKit | |
| Number of saturated rings | RDKit | |
| Number of Smallest Set of Smallest Rings (SSSR) | RDKit | |