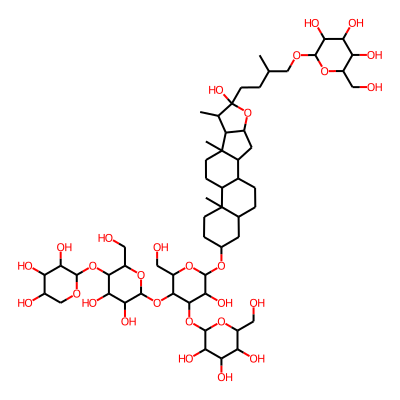
Summary
SMILES: OCC1OC(OC2CCC3(C(C2)CCC2C3CCC3(C2CC2C3C(C)C(O2)(O)CCC(COC2OC(CO)C(C(C2O)O)O)C)C)C)C(C(C1OC1OC(CO)C(C(C1O)O)OC1OCC(C(C1O)O)O)OC1OC(CO)C(C(C1O)O)O)OInChI: InChI=1S/C56H94O28/c1-21(19-74-49-42(69)38(65)36(63)30(15-57)77-49)7-12-56(73)22(2)34-29(84-56)14-27-25-6-5-23-13-24(8-10-54(23,3)26(25)9-11-55(27,34)4)76-53-45(72)48(83-51-43(70)39(66)37(64)31(16-58)78-51)47(33(18-60)80-53)82-52-44(71)40(67)46(32(17-59)79-52)81-50-41(68)35(62)28(61)20-75-50/h21-53,57-73H,5-20H2,1-4H3InChIKey: SXCDJKBVZMMBQG-UHFFFAOYSA-N
DeepSMILES: OCCOCOCCCCCC6)CCCC6CCCC6CCC5CC)CO5)O)CCCCOCOCCO))CCC6O))O))O)))))))C))))))))))C)))))))))C))))))CCC6OCOCCO))CCC6O))O))OCOCCCC6O))O))O))))))))))))OCOCCO))CCC6O))O))O)))))))O
Scaffold Graph/Node/Bond level: C(CCC1CC2C(CC3C2CCC2C4CCC(OC5CC(OC6CCCCO6)C(OC6CCC(OC7CCCCO7)CO6)CO5)CC4CCC23)O1)COC1CCCCO1
Scaffold Graph/Node level: C(CCC1CC2C(CC3C2CCC2C4CCC(OC5CC(OC6CCCCO6)C(OC6CCC(OC7CCCCO7)CO6)CO5)CC4CCC23)O1)COC1CCCCO1
Scaffold Graph level: C1CCC(CCCCCC2CC3CC4C(CCC5C6CCC(CC7CCC(CC8CCC(CC9CCCCC9)CC8)C(CC8CCCCC8)C7)CC6CCC54)C3C2)CC1
Functional groups: CO; COC(C)(C)O; COC(C)OC
Chemical classification
ClassyFire Kingdom: Organic compounds
ClassyFire Superclass: Lipids and lipid-like moleculesClassyFire Class: Steroids and steroid derivatives
ClassyFire Subclass: Steroidal glycosides
NP Classifier Biosynthetic pathway: Terpenoids
NP Classifier Superclass: Steroids
NP Classifier Class: Furostane steroids
Synonymous chemical names:asparagoside h
External chemical identifiers:CID:3042723
Chemical structure download