Summary
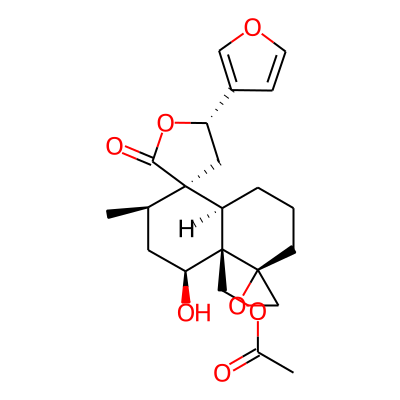
SMILES: CC(=O)OC[C@@]12[C@@H](O)C[C@H]([C@@]3([C@H]1CCC[C@@]12CO1)C[C@H](OC3=O)c1cocc1)CInChI: InChI=1S/C22H28O7/c1-13-8-18(24)22(12-27-14(2)23)17(4-3-6-20(22)11-28-20)21(13)9-16(29-19(21)25)15-5-7-26-10-15/h5,7,10,13,16-18,24H,3-4,6,8-9,11-12H2,1-2H3/t13-,16+,17-,18+,20+,21-,22+/m1/s1InChIKey: LOZYVHYKXUKJDA-UYUQIPAXSA-N
DeepSMILES: CC=O)OC[C@@][C@@H]O)C[C@H][C@@][C@H]6CCC[C@]%10CO3)))))))C[C@H]OC5=O)))ccocc5))))))))C
Scaffold Graph/Node/Bond level: O=C1OC(c2ccoc2)CC12CCCC1C2CCCC12CO2
Scaffold Graph/Node level: OC1OC(C2CCOC2)CC12CCCC1C2CCCC12CO2
Scaffold Graph level: CC1CC(C2CCCC2)CC12CCCC1C2CCCC12CC2
Functional groups: CO; COC(C)=O; C[C@@]1(C)CO1; coc
Chemical classification
ClassyFire Kingdom: Organic compounds
ClassyFire Superclass: Organoheterocyclic compoundsClassyFire Class: Lactones
ClassyFire Subclass: Gamma butyrolactones
NP Classifier Biosynthetic pathway: Terpenoids
NP Classifier Superclass: Diterpenoids
NP Classifier Class: Colensane and Clerodane diterpenoids
Synonymous chemical names:teupolin i
External chemical identifiers:CID:156030; ZINC:ZINC000005037505
Chemical structure download