Summary
IMPPAT Phytochemical identifier: IMPHY002323
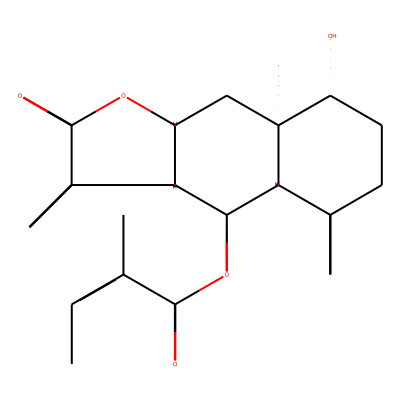
Phytochemical name: Chrysanin
Synonymous chemical names:chrysanin
Chemical structure information
SMILES:C/C=C(C(=O)O[C@H]1[C@H]2[C@@H](OC(=O)C2=C)C[C@@]2([C@@H]1C(=C)CC[C@H]2O)C)/CInChI:InChI=1S/C20H26O5/c1-6-10(2)18(22)25-17-15-12(4)19(23)24-13(15)9-20(5)14(21)8-7-11(3)16(17)20/h6,13-17,21H,3-4,7-9H2,1-2,5H3/b10-6-/t13-,14+,15+,16+,17-,20-/m0/s1InChIKey:CCJPWAPOFONQQT-KMJRVMNLSA-N
DeepSMILES:C/C=CC=O)O[C@H][C@H][C@@H]OC=O)C5=C))))C[C@@][C@@H]6C=C)CC[C@H]6O))))))C))))))))/C
Functional groups:C/C=C(/C)C(=O)OC, C=C(C)C, C=C1CCOC1=O, CO
Molecular scaffolds
Scaffold Graph/Node/Bond level:C=C1CCCC2CC3OC(=O)C(=C)C3CC12
Scaffold Graph/Node level:CC1CCCC2CC3OC(O)C(C)C3CC12
Scaffold Graph level:CC1CC2CC3CCCC(C)C3CC2C1C
Chemical classification
ClassyFire Kingdom: Organic compounds
ClassyFire Superclass: Lipids and lipid-like moleculesClassyFire Class: Prenol lipids
ClassyFire Subclass: Terpene lactones
NP Classifier Biosynthetic pathway: Terpenoids
NP Classifier Superclass: Sesquiterpenoids
NP Classifier Class: Eudesmane sesquiterpenoids
NP-Likeness score: 3.665
Chemical structure download