Summary
IMPPAT Phytochemical identifier: IMPHY003573
Phytochemical name: Reticuline
Synonymous chemical names:(+)-reticuline, (+)reticuline, (s)-reticuline, +-reticuline, l(+)-reticuline, reticuline, reticuline, (+), reticuline,(+)
External chemical identifiers:CID:439653, ChEMBL:CHEMBL235212, ChEBI:16718, ZINC:ZINC000000895630, FDASRS:X35Z551WT4, SureChEMBL:SCHEMBL147597, MolPort-005-981-212
Chemical structure information
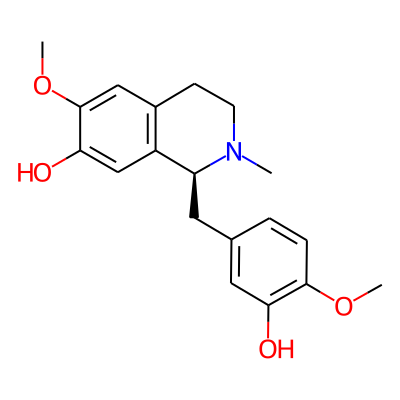
SMILES:
COc1ccc(cc1O)C[C@@H]1N(C)CCc2c1cc(O)c(c2)OCInChI:
InChI=1S/C19H23NO4/c1-20-7-6-13-10-19(24-3)17(22)11-14(13)15(20)8-12-4-5-18(23-2)16(21)9-12/h4-5,9-11,15,21-22H,6-8H2,1-3H3/t15-/m0/s1InChIKey:
BHLYRWXGMIUIHG-HNNXBMFYSA-NDeepSMILES:
COcccccc6O)))C[C@@H]NC)CCcc6ccO)cc6)OCFunctional groups:
CN(C)C, cO, cOC
Molecular scaffolds
Scaffold Graph/Node/Bond level:
c1ccc(CC2NCCc3ccccc32)cc1Scaffold Graph/Node level:
C1CCC(CC2NCCC3CCCCC32)CC1Scaffold Graph level:
C1CCC(CC2CCCC3CCCCC32)CC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Organoheterocyclic compoundsClassyFire Class: Isoquinolines and derivatives
ClassyFire Subclass: Benzylisoquinolines
NP Classifier Biosynthetic pathway: Alkaloids
NP Classifier Superclass: Tyrosine alkaloids
NP Classifier Class: Isoquinoline alkaloids, Tetrahydroisoquinoline alkaloids
NP-Likeness score: 1.071
Chemical structure download