Summary
IMPPAT Phytochemical identifier: IMPHY004470
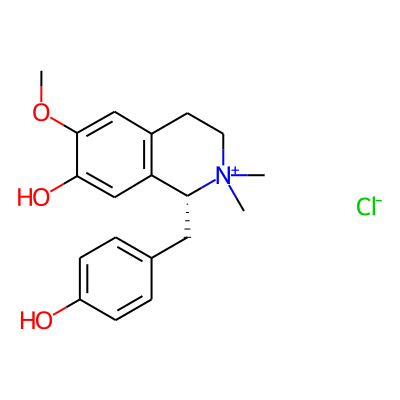
Phytochemical name: Isoquinolinium, 1,2,3,4-tetrahydro-7-hydroxy-1-((4-hydroxyphenyl)methyl)-6-methoxy-2,2-dimethyl-, chloride, (R)-
Synonymous chemical names:magnocurarine chloride
External chemical identifiers:CID:53265
Chemical structure information
SMILES:
COc1cc2CC[N+]([C@@H](c2cc1O)Cc1ccc(cc1)O)(C)C.[Cl-]InChI:
InChI=1S/C19H23NO3.ClH/c1-20(2)9-8-14-11-19(23-3)18(22)12-16(14)17(20)10-13-4-6-15(21)7-5-13;/h4-7,11-12,17H,8-10H2,1-3H3,(H-,21,22);1H/t17-;/m1./s1InChIKey:
VPFXIJWKEYUDJZ-UNTBIKODSA-NDeepSMILES:
COcccCC[N+][C@@H]c6cc%10O))))Ccccccc6))O)))))))C)C.[Cl-]Functional groups:
C[N+](C)(C)C, [Cl-], cO, cOC
Molecular scaffolds
Scaffold Graph/Node/Bond level:
c1ccc(CC2[NH2+]CCc3ccccc32)cc1Scaffold Graph/Node level:
C1CCC(CC2NCCC3CCCCC32)CC1Scaffold Graph level:
C1CCC(CC2CCCC3CCCCC32)CC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Organoheterocyclic compoundsClassyFire Class: Isoquinolines and derivatives
ClassyFire Subclass: Benzylisoquinolines
NP Classifier Biosynthetic pathway: Alkaloids
NP Classifier Superclass: Tyrosine alkaloids
NP Classifier Class: Isoquinoline alkaloids
NP-Likeness score: 1.592
Chemical structure download