Summary
IMPPAT Phytochemical identifier: IMPHY006301
Phytochemical name: Tubocurarine chloride
Synonymous chemical names:d-tubocurarine, tubocurarine
External chemical identifiers:CID:6000, ChEMBL:CHEMBL339427, ChEBI:9774, ZINC:ZINC000003978083, FDASRS:W9YXS298BM, SureChEMBL:SCHEMBL121375
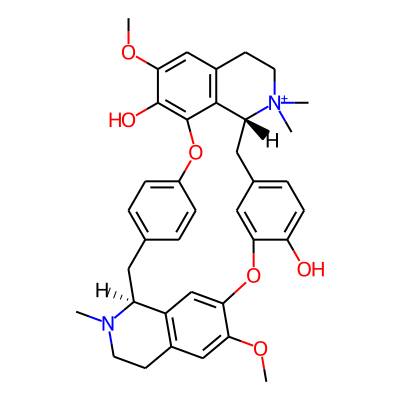
Chemical structure information
SMILES:
COc1cc2CCN([C@@H]3c2cc1Oc1cc(ccc1O)C[C@@H]1c2c(CC[N+]1(C)C)cc(c(c2Oc1ccc(C3)cc1)O)OC)CInChI:
InChI=1S/C37H40N2O6/c1-38-14-12-24-19-32(42-4)33-21-27(24)28(38)16-22-6-9-26(10-7-22)44-37-35-25(20-34(43-5)36(37)41)13-15-39(2,3)29(35)17-23-8-11-30(40)31(18-23)45-33/h6-11,18-21,28-29H,12-17H2,1-5H3,(H-,40,41)/p+1/t28-,29+/m0/s1InChIKey:
JFJZZMVDLULRGK-URLMMPGGSA-ODeepSMILES:
COcccCCN[C@@H]c6cc%10Occcccc6O))))C[C@@H]ccCC[N+]6C)C))))cccc6OccccC%22)cc6))))))))O))OC))))))))))))))))CFunctional groups:
CN(C)C, C[N+](C)(C)C, cO, cOC, cOc
Molecular scaffolds
Scaffold Graph/Node/Bond level:
c1cc2cc(c1)Oc1ccc3c(c1)C(Cc1ccc(cc1)Oc1cccc4c1C(C2)[NH2+]CC4)NCC3Scaffold Graph/Node level:
C1CC2CC(C1)OC1CCC3CCNC(CC4CCC(CC4)OC4CCCC5CCNC(C2)C54)C3C1Scaffold Graph level:
C1CC2CC(C1)CC1CCCC3CCCC(CC4CCC(CC4)CC4CCCC5CCC(C2)CC54)C31
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Organic oxygen compoundsClassyFire Class: Organooxygen compounds
ClassyFire Subclass: Ethers
NP Classifier Biosynthetic pathway: Alkaloids
NP Classifier Superclass: Tyrosine alkaloids
NP Classifier Class: Tetrahydroisoquinoline alkaloids, Isoquinoline alkaloids
NP-Likeness score: 2.261
Chemical structure download