Summary
IMPPAT Phytochemical identifier: IMPHY006485
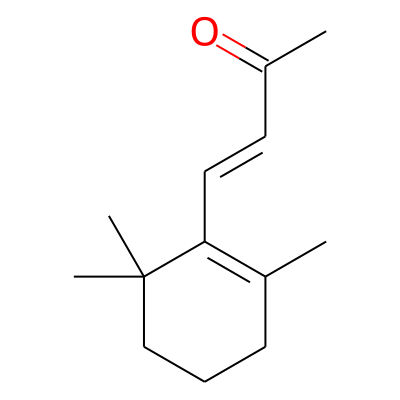
Phytochemical name: beta-Ionone
Synonymous chemical names:(e )- β -ionone, (e)- β -ionone, (e)- β-ionone, (e)-b-ionone, (e)-p-ionone, (e)-β -ionone, (e)-β- ionone, (e)-β-ionene, (e)-β-ionone, beta ionone, beta-ionone, beta-ionone*, e- β -ionone, e-β-lonone, trans-β-ionone, â-ionone*, β -ionone, β- ionone, β-ionone, β-ionone*, β-ionones
External chemical identifiers:CID:638014, ChEMBL:CHEMBL559945, ChEBI:32325, ZINC:ZINC000003881456, FDASRS:A7NRR1HLH6, SureChEMBL:SCHEMBL23953, MolPort-001-783-113
Chemical structure information
SMILES:
CC(=O)/C=C/C1=C(C)CCCC1(C)CInChI:
InChI=1S/C13H20O/c1-10-6-5-9-13(3,4)12(10)8-7-11(2)14/h7-8H,5-6,9H2,1-4H3/b8-7+InChIKey:
PSQYTAPXSHCGMF-BQYQJAHWSA-NDeepSMILES:
CC=O)/C=C/C=CC)CCCC6C)CFunctional groups:
CC(=O)/C=C/C(C)=C(C)C
Molecular scaffolds
Scaffold Graph/Node/Bond level:
C1=CCCCC1Scaffold Graph/Node level:
C1CCCCC1Scaffold Graph level:
C1CCCCC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Lipids and lipid-like moleculesClassyFire Class: Prenol lipids
ClassyFire Subclass: Sesquiterpenoids
NP Classifier Biosynthetic pathway: Terpenoids
NP Classifier Superclass: Apocarotenoids
NP Classifier Class: Apocarotenoids (β-), Megastigmanes
NP-Likeness score: 2.09
Chemical structure download