Summary
IMPPAT Phytochemical identifier: IMPHY007379
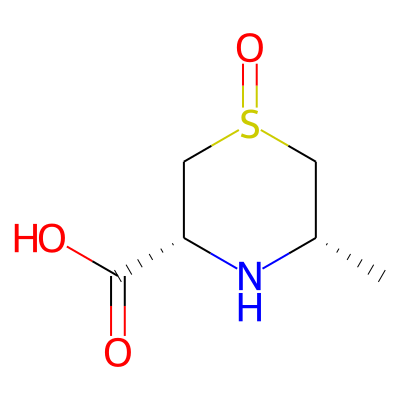
Phytochemical name: (3R,5S)-5-methyl-1-oxo-1,4-thiazinane-3-carboxylic acid
Synonymous chemical names:cyclo alliin, cycloalliin, cycloallin
External chemical identifiers:CID:91808874
Chemical structure information
SMILES:
C[C@H]1CS(=O)C[C@H](N1)C(=O)OInChI:
InChI=1S/C6H11NO3S/c1-4-2-11(10)3-5(7-4)6(8)9/h4-5,7H,2-3H2,1H3,(H,8,9)/t4-,5-,11?/m0/s1InChIKey:
JYMHODZXTIGVPA-OHYCWAGJSA-NDeepSMILES:
C[C@H]CS=O)C[C@H]N6)C=O)OFunctional groups:
CC(=O)O, CNC, CS(C)=O
Molecular scaffolds
Scaffold Graph/Node/Bond level:
O=S1CCNCC1Scaffold Graph/Node level:
O[SH]1CCNCC1Scaffold Graph level:
CC1CCCCC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Organic acids and derivativesClassyFire Class: Carboxylic acids and derivatives
ClassyFire Subclass: Amino acids, peptides, and analogues
NP Classifier Biosynthetic pathway: Amino acids and Peptides
NP Classifier Superclass: Small peptides
NP Classifier Class: Aminoacids
NP-Likeness score: 0.985
Chemical structure download