Summary
IMPPAT Phytochemical identifier: IMPHY010896
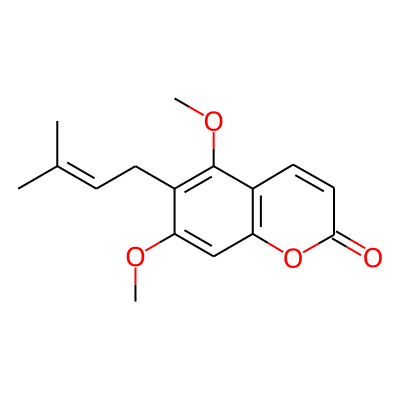
Phytochemical name: 2H-1-Benzopyran-2-one, 5,7-dimethoxy-6-(3-methyl-2-butenyl)-
Synonymous chemical names:toddaculin, toddaculine[5,7-dimethoxy-6-(2-isopentenyl)-coumarin]
External chemical identifiers:CID:5321960, ChEMBL:CHEMBL3235996, SureChEMBL:SCHEMBL15941538, MolPort-044-754-147
Chemical structure information
SMILES:
COc1cc2oc(=O)ccc2c(c1CC=C(C)C)OCInChI:
InChI=1S/C16H18O4/c1-10(2)5-6-11-13(18-3)9-14-12(16(11)19-4)7-8-15(17)20-14/h5,7-9H,6H2,1-4H3InChIKey:
KRQHZFHWEAJPNO-UHFFFAOYSA-NDeepSMILES:
COcccoc=O)ccc6cc%10CC=CC)C)))))OCFunctional groups:
CC=C(C)C, c=O, cOC, coc
Molecular scaffolds
Scaffold Graph/Node/Bond level:
O=c1ccc2ccccc2o1Scaffold Graph/Node level:
OC1CCC2CCCCC2O1Scaffold Graph level:
CC1CCC2CCCCC2C1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Phenylpropanoids and polyketidesClassyFire Class: Coumarins and derivatives
NP Classifier Biosynthetic pathway: Shikimates and Phenylpropanoids
NP Classifier Superclass: Coumarins
NP Classifier Class: Simple coumarins
NP-Likeness score: 1.713
Chemical structure download