Summary
IMPPAT Phytochemical identifier: IMPHY011511
Phytochemical name: Lappaol E
Synonymous chemical names:lappaol e
External chemical identifiers:CID:73425454, FDASRS:8882OGD76K
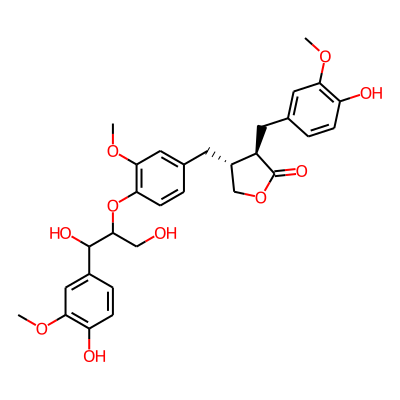
Chemical structure information
SMILES:
COc1cc(ccc1OC(C(c1ccc(c(c1)OC)O)O)CO)C[C@H]1COC(=O)[C@@H]1Cc1ccc(c(c1)OC)OInChI:
InChI=1S/C30H34O10/c1-36-25-12-18(4-7-22(25)32)11-21-20(16-39-30(21)35)10-17-5-9-24(27(13-17)38-3)40-28(15-31)29(34)19-6-8-23(33)26(14-19)37-2/h4-9,12-14,20-21,28-29,31-34H,10-11,15-16H2,1-3H3/t20-,21+,28?,29?/m0/s1InChIKey:
ASYBYLYCBSRGRZ-KWCNVUSUSA-NDeepSMILES:
COcccccc6OCCcccccc6)OC)))O)))))O))CO)))))))C[C@H]COC=O)[C@@H]5Ccccccc6)OC)))OFunctional groups:
CO, COC(C)=O, cO, cOC
Molecular scaffolds
Scaffold Graph/Node/Bond level:
O=C1OCC(Cc2ccc(OCCc3ccccc3)cc2)C1Cc1ccccc1Scaffold Graph/Node level:
OC1OCC(CC2CCC(OCCC3CCCCC3)CC2)C1CC1CCCCC1Scaffold Graph level:
CC1CCC(CC2CCC(CCCC3CCCCC3)CC2)C1CC1CCCCC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Lignans, neolignans and related compoundsClassyFire Class: Furanoid lignans
ClassyFire Subclass: Tetrahydrofuran lignans
NP Classifier Biosynthetic pathway: Shikimates and Phenylpropanoids
NP Classifier Superclass: Lignans
NP Classifier Class: Dibenzylbutyrolactone lignans, Neolignans
NP-Likeness score: 1.336
Chemical structure download