Summary
IMPPAT Phytochemical identifier: IMPHY013042
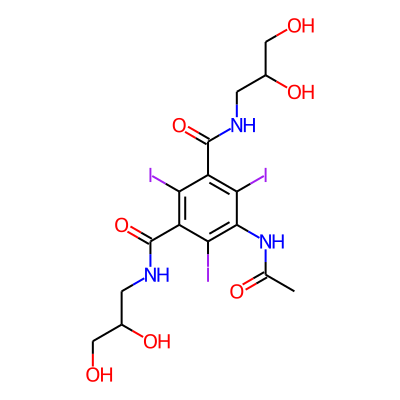
Phytochemical name: 5-(Acetylamino)-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
Synonymous chemical names:hydrolysate
External chemical identifiers:CID:11115326, FDASRS:BK2UH2P436, SureChEMBL:SCHEMBL619944, MolPort-003-844-389
Chemical structure information
SMILES:
OCC(CNC(=O)c1c(I)c(C(=O)NCC(CO)O)c(c(c1I)NC(=O)C)I)OInChI:
InChI=1S/C16H20I3N3O7/c1-6(25)22-14-12(18)9(15(28)20-2-7(26)4-23)11(17)10(13(14)19)16(29)21-3-8(27)5-24/h7-8,23-24,26-27H,2-5H2,1H3,(H,20,28)(H,21,29)(H,22,25)InChIKey:
BHCBLTRDEYPMFZ-UHFFFAOYSA-NDeepSMILES:
OCCCNC=O)ccI)cC=O)NCCCO))O)))))ccc6I))NC=O)C))))I))))))))OFunctional groups:
CO, cC(=O)NC, cI, cNC(C)=O
Molecular scaffolds
Scaffold Graph/Node/Bond level:
c1ccccc1Scaffold Graph/Node level:
C1CCCCC1Scaffold Graph level:
C1CCCCC1
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: BenzenoidsClassyFire Class: Benzene and substituted derivatives
ClassyFire Subclass: Benzoic acids and derivatives
NP Classifier Biosynthetic pathway: Alkaloids
NP Classifier Superclass: Peptide alkaloids
NP Classifier Class: Simple amide alkaloids
NP-Likeness score: -0.266
Chemical structure download