Summary
IMPPAT Phytochemical identifier: IMPHY013945
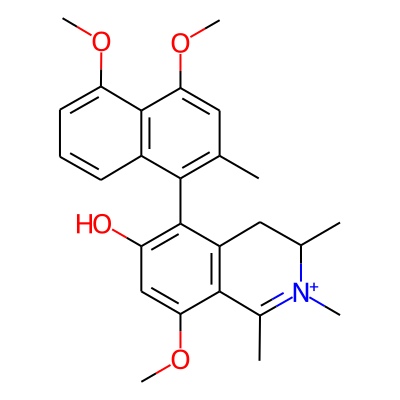
Phytochemical name: 5-(4,5-Dimethoxy-2-methylnaphthalen-1-yl)-8-methoxy-1,2,3-trimethyl-3,4-dihydroisoquinolin-2-ium-6-ol
Synonymous chemical names:ancistrocladinine
External chemical identifiers:CID:375959
Chemical structure information
SMILES:
COc1cc(O)c(c2c1C(=[N+](C)C(C2)C)C)c1c(C)cc(c2c1cccc2OC)OCInChI:
InChI=1S/C26H29NO4/c1-14-11-21(30-6)26-17(9-8-10-20(26)29-5)23(14)25-18-12-15(2)27(4)16(3)24(18)22(31-7)13-19(25)28/h8-11,13,15H,12H2,1-7H3/p+1InChIKey:
APRMONYQVUUJSI-UHFFFAOYSA-ODeepSMILES:
COcccO)ccc6C=[N+]C)CC6)C)))C))))ccC)cccc6cccc6OC))))))))OCFunctional groups:
cC(C)=[N+](C)C, cO, cOC
Molecular scaffolds
Scaffold Graph/Node/Bond level:
C1=[NH+]CCc2c1cccc2-c1cccc2ccccc12Scaffold Graph/Node level:
C1CCC2C(C1)CCCC2C1CCCC2CNCCC21Scaffold Graph level:
C1CCC2C(C1)CCCC2C1CCCC2CCCCC21
Chemical classification
ClassyFire Kingdom: Organic compoundsClassyFire Superclass: Organoheterocyclic compoundsClassyFire Class: Isoquinolines and derivatives
ClassyFire Subclass: Naphthylisoquinolines
NP Classifier Biosynthetic pathway: Alkaloids
NP Classifier Superclass: Tyrosine alkaloids
NP Classifier Class: Isoquinoline alkaloids
NP-Likeness score: 1.133
Chemical structure download